Respiration is a vital life process that releases energy from food. Every cell in the body performs this process continuously. But many students ask, why is respiration considered an exothermic reaction? This question is important for the NCERT Class 10 exams. It also helps you understand how your body works daily.
An exothermic reaction releases energy in the form of heat. During respiration, glucose breaks down and releases energy. This energy supports movement, growth, and body functions. Let’s understand this concept step by step.

What Is an Exothermic Reaction?
An exothermic reaction is a chemical reaction that releases heat energy. The energy flows from the system to the surroundings. As a result, the surroundings may feel warm.
For example, burning wood releases heat and light. Similarly, combustion of fuels produces energy. These are common exothermic reaction examples. Therefore, any reaction that gives out energy is called exothermic.

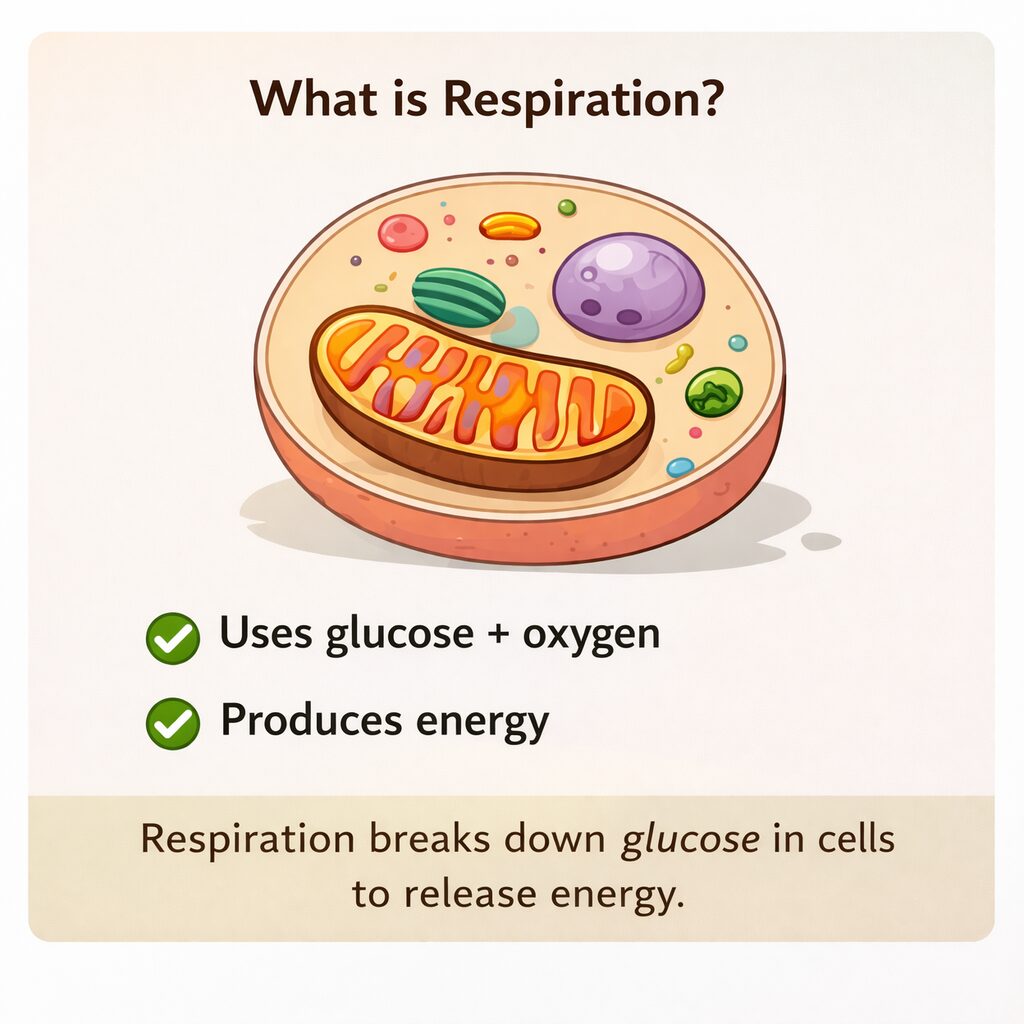
What Is Respiration?
Respiration is the process by which cells break down food. It mainly uses glucose and oxygen. This process occurs inside the mitochondria of cells.
There are two main types of respiration. Aerobic respiration uses oxygen and releases more energy. Anaerobic respiration occurs without oxygen and releases less energy. However, both forms release energy. Hence, respiration is always linked with energy production.
Why Is Respiration Considered an Exothermic Reaction?
Respiration is considered an exothermic reaction because it releases energy. During this process, glucose molecules break down into simpler substances. This breakdown releases stored chemical energy.
Moreover, part of this energy is released as heat. This heat helps maintain body temperature. The remaining energy is stored in ATP molecules. Therefore, respiration clearly shows the characteristics of an exothermic reaction.
In simple terms, energy comes out during respiration. That is why respiration is an exothermic process.
Chemical Equation of Respiration
The respiration reaction can be written as:
C₆H₁₂O₆ + 6O₂ → 6CO₂ + 6H₂O + Energy
This equation shows that glucose reacts with oxygen. It produces carbon dioxide, water, and energy. The release of energy proves that respiration is an exothermic reaction.
Furthermore, this energy is essential for all living activities. Without it, the body cannot function properly.
How Energy Is Released in Respiration
First, glucose enters the cell and breaks down into smaller molecules. This process happens in several steps. Each step releases a small amount of energy.
Next, this energy is stored in ATP molecules. ATP acts as the energy currency of the cell. It powers various biological functions. At the same time, some energy is lost as heat.
Therefore, respiration not only produces usable energy but also releases heat. This confirms its exothermic reaction nature.
Real-Life Examples of the Exothermic Nature of Respiration
Respiration shows its effects in daily life. For example, your body stays warm even in cold weather. This warmth comes from heat released during respiration.
During exercise, your body produces more energy. As a result, you feel hot and start sweating. This happens because the respiration rate increases.
Thus, these examples clearly show that respiration releases heat. Hence, it is an exothermic reaction.
Is Respiration Exothermic or Endothermic?
Students often confuse this concept. However, the answer is simple. Respiration is always an exothermic reaction.
In contrast, endothermic reactions absorb energy. For example, photosynthesis absorbs sunlight energy. Respiration does the opposite. It releases energy instead of absorbing it.
Therefore, respiration cannot be endothermic. It is purely exothermic in nature.
Why is This Important for Class 10 Students?
This question is frequently asked in exams. Students should answer it in simple and clear words.
You can write:
Respiration is an exothermic reaction because it releases energy during the breakdown of glucose. This energy is released as heat and stored in ATP.
This answer is short, accurate, and exam-friendly.
Common Exam Questions
Q1: Why is respiration considered an exothermic reaction?
Respiration releases energy when glucose breaks down. Therefore, it is an exothermic reaction.
Q2: Is respiration exothermic or endothermic?
Respiration is an exothermic reaction because it releases heat energy.
Q3: Write the respiration reaction.
C₆H₁₂O₆ + 6O₂ → 6CO₂ + 6H₂O + Energy.
Frequently Asked Questions
Respiration is considered an exothermic reaction because it releases energy when glucose breaks down in the presence of oxygen. This energy is partly released as heat and partly stored in ATP, which supports essential body functions like movement and growth.
Yes, respiration is always an exothermic reaction because it consistently releases energy during the breakdown of food molecules. Whether aerobic or anaerobic, the process produces energy, ensuring that cells receive power for metabolic activities and survival.
Respiration is a biochemical and exothermic reaction that occurs inside living cells. It involves the breakdown of glucose using oxygen to release energy, forming carbon dioxide and water as byproducts, while also generating ATP for cellular processes.
Respiration releases heat because the chemical energy stored in glucose gets converted during its breakdown. Not all energy converts into ATP, so excess energy escapes as heat, helping maintain body temperature and supporting metabolic balance in living organisms.